Description
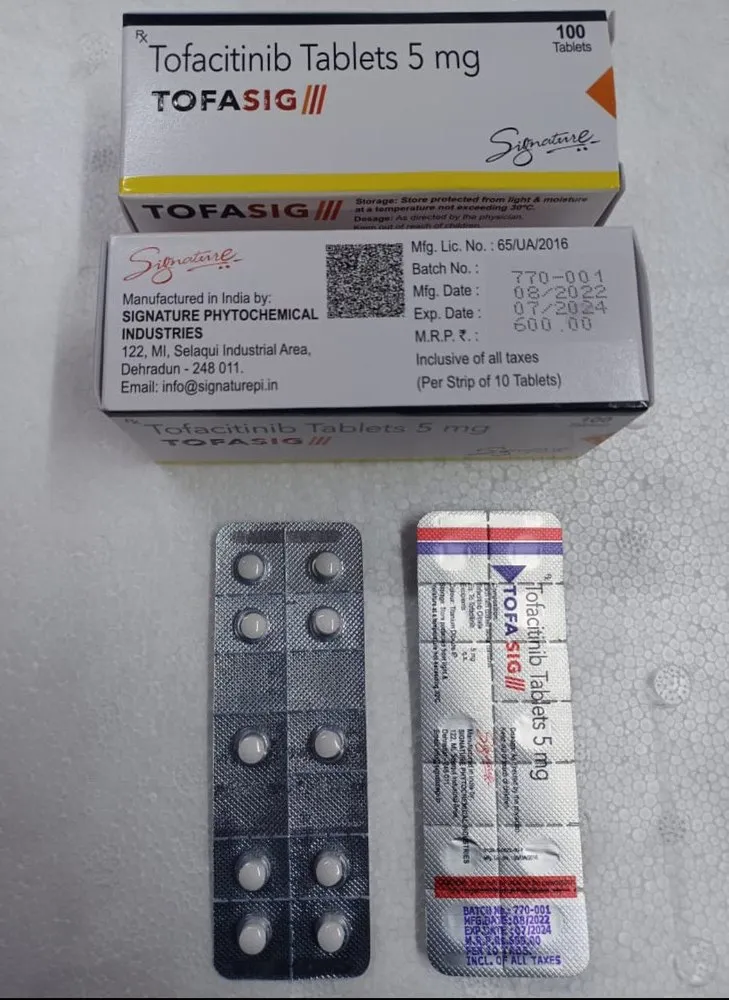
TOFASIG TABLET contains Tofacitinib, an advanced Janus kinase (JAK) inhibitor, used in the treatment of moderate to severe autoimmune diseases. By inhibiting the JAK-STAT signaling pathway, TOFASIG helps regulate the immune system and reduce symptoms such as joint pain, swelling, and stiffness.
It is commonly prescribed for patients with rheumatoid arthritis (RA), psoriatic arthritis (PsA), and ulcerative colitis (UC) who have had an inadequate response to other conventional therapies like methotrexate or TNF inhibitors.
Janus Kinase (JAK) Inhibitor – Disease-Modifying Anti-Rheumatic Drug (DMARD)
Tofacitinib selectively inhibits JAK enzymes (primarily JAK1 and JAK3), which play a crucial role in cytokine signaling within the immune system. By blocking these enzymes, TOFASIG reduces inflammation and halts immune-mediated damage to joints and tissues.
Rheumatoid Arthritis (RA)
Psoriatic Arthritis (PsA)
Ulcerative Colitis (UC)
Other inflammatory autoimmune diseases (as per physician guidance)
Reduces joint inflammation, stiffness, and pain
Prevents progression of autoimmune damage
Improves physical function and quality of life
Oral administration offers convenience over injectables
Can be used in patients intolerant to biologics
Commonly prescribed as 5 mg twice daily or extended-release formulations once daily, depending on the indication
Must be taken exactly as directed by the physician
Monitoring required for blood counts, liver enzymes, and lipid levels
Not recommended during pregnancy or breastfeeding unless prescribed
Regular screening for infections (e.g., TB, hepatitis) is required before starting
Use with caution in patients with liver or kidney disease
Can increase risk of serious infections, blood clots, or malignancy with long-term use
Upper respiratory infections
Headache, diarrhea
Increased liver enzymes
High cholesterol levels
Risk of blood clots and serious infections in rare cases
BLUEPILL EXPRESS is a globally trusted third-party manufacturer and exporter of high-standard medicines. TOFASIG TABLET is developed under rigorous GMP guidelines and is tested for consistency, potency, and safety.
We offer:
WHO-GMP & ISO-certified production
Custom branding & private labeling services
Regulatory documentation for global markets
Bulk export to over 50 countries
TOFASIG TABLET is part of our expanding global portfolio, exported to healthcare providers and distributors in:
Latin America
Southeast Asia
Africa & the Middle East
CIS Countries
Europe & Oceania
We ensure timely delivery, proper documentation, and high-quality compliance in all international transactions.