Description
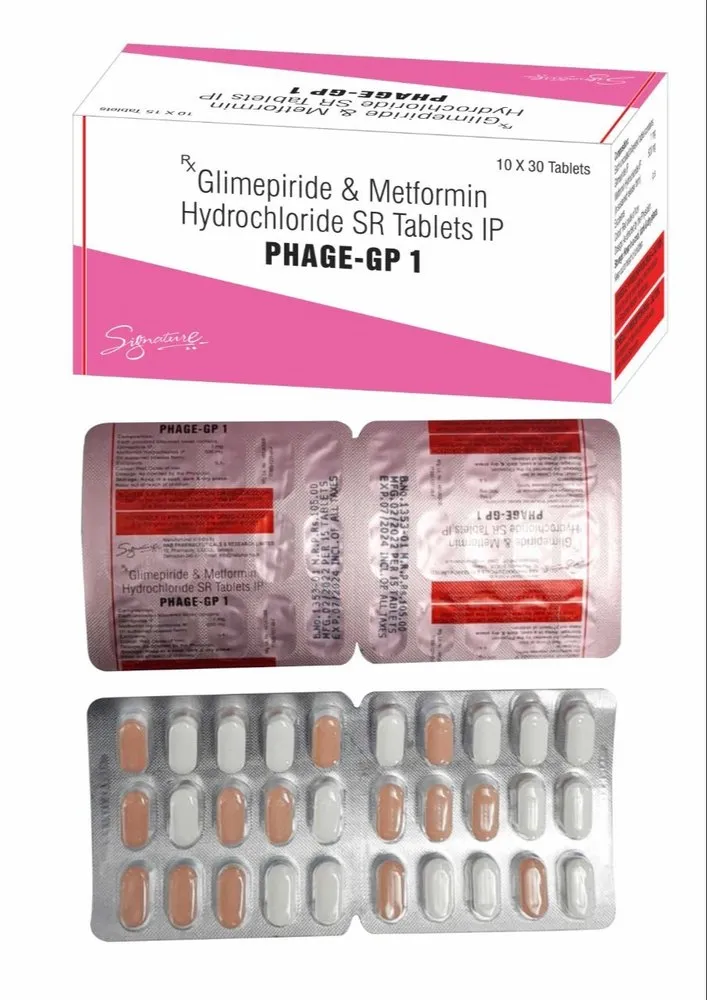
PHAGE GP 1 TABLET is an oral antidiabetic combination medication that contains Glimepiride 1 mg and Metformin Hydrochloride (usually 500 mg, either immediate-release or sustained-release). This fixed-dose combination is widely prescribed for patients with Type 2 Diabetes Mellitus who require more than one oral agent to achieve optimal blood glucose control.
Glimepiride, a sulfonylurea, stimulates pancreatic beta cells to release insulin, thus reducing blood glucose levels.
Metformin, a biguanide, decreases hepatic glucose production and improves peripheral insulin sensitivity.
Lower fasting and postprandial blood glucose levels
Improve glycemic control in patients unresponsive to monotherapy
Reduce the risk of diabetes-related complications
Type 2 Diabetes Mellitus (when diet, exercise, and monotherapy are inadequate)
First-line or step-up therapy in diabetic patients
Long-term blood sugar regulation and metabolic control
Combination of two complementary antidiabetic agents
Helps reduce insulin resistance and boost insulin secretion
Well-tolerated with a reduced risk of hypoglycemia compared to older sulfonylureas
Enhances patient adherence by simplifying therapy into a single tablet
Proven to reduce HbA1c over time
PHAGE GP 1 TABLET is manufactured in WHO-GMP, ISO, and FDA-compliant pharmaceutical facilities. It undergoes rigorous quality checks to ensure uniformity, potency, and therapeutic efficacy.
BLUEPILL EXPRESS, the global distributor of PHAGE GP 1 TABLET, is recognized for its world-class pharmaceutical exports. Serving regions across North America, Europe, Africa, Asia, the Middle East, and Latin America, BLUEPILL EXPRESS is a leading name in third-party and contract manufacturing of high-quality generic and branded formulations.
Adult patients with Type 2 Diabetes Mellitus
Individuals transitioning from monotherapy to combination therapy
Patients aiming for long-term glycemic and metabolic control